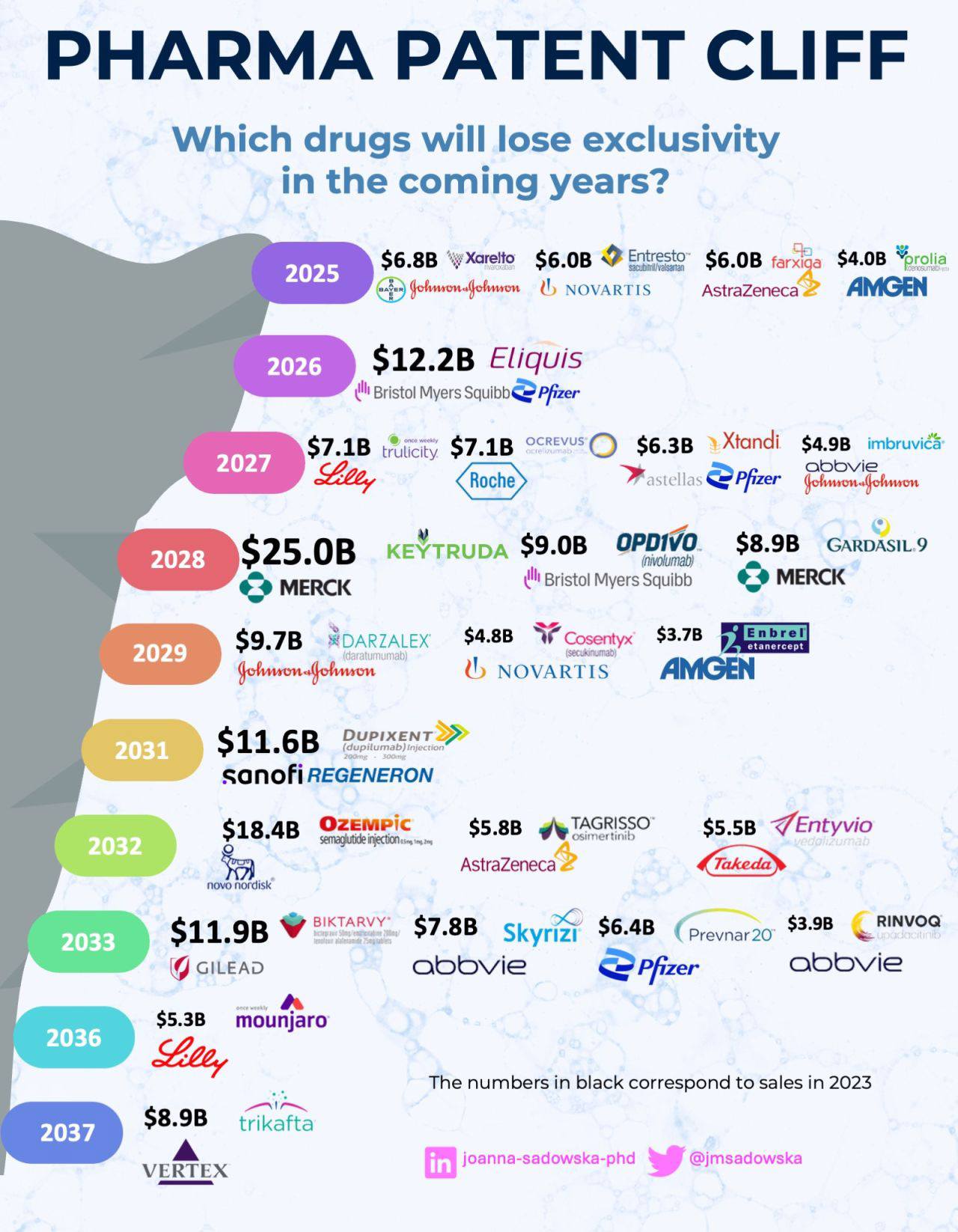
Pharmaceutical companies do not lose billions when a patent expires. They lose billions in the years before the patent expires, when marketing restrictions, compliance risk, pricing pressure, and payer negotiations begin to tighten around the brand. By the time generic competition officially enters the market, revenue decline has already started. The real battle happens during the transition period, when companies still hold patent protection but face growing restrictions on what they can say, how they can market, and how they can defend market share.
If you work in pharmaceutical marketing or commercial strategy, the patent cliff is not a legal event. It is a marketing and market access crisis that unfolds over several years. During this period, every marketing claim, lifecycle strategy, pricing decision, and physician engagement program faces higher scrutiny from regulators, payers, and competitors preparing generic entry.
This is the most dangerous phase in a drug’s commercial life, and most marketing teams are not trained for it.
What the Patent Cliff Really Means for Marketing Teams
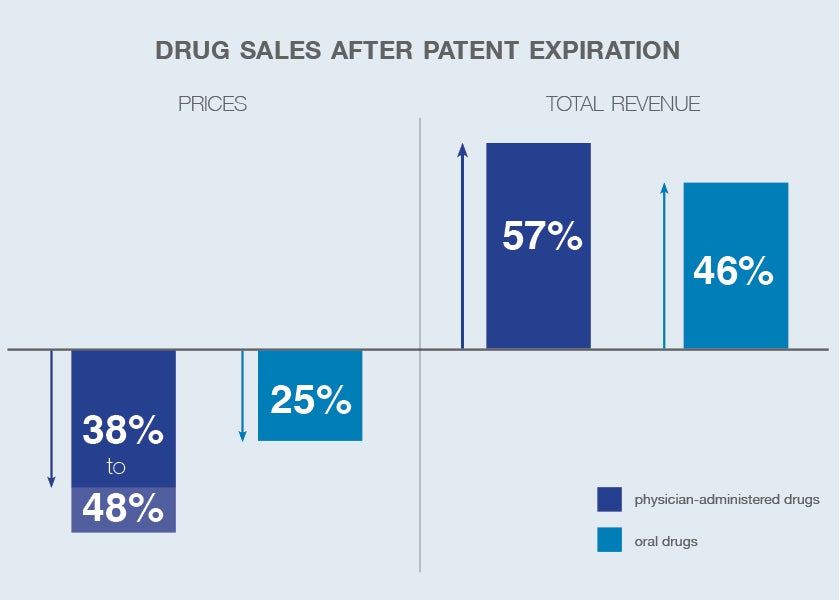
When a pharmaceutical patent expires, generic competitors can enter the market and sell the same drug at significantly lower prices. In many cases, drug prices drop by 70 to 90 percent within the first year of generic entry. Market share for the original brand often collapses rapidly.
Blockbuster drugs that once generated billions in annual revenue can lose most of their market share within 12 to 24 months after generic entry. This sudden revenue decline is known as the patent cliff.
Some of the most well-known patent cliff examples include:
- Lipitor lost billions in revenue after patent expiration
- Plavix faced rapid generic competition after exclusivity ended
- Humira began facing biosimilar competition after years of patent protection
- Advair lost market share after generic competition entered respiratory markets
These examples show that the patent cliff is not a gradual decline. It is often a sharp revenue drop.
For marketing teams, this creates a difficult situation. You must continue promoting the brand while preparing for revenue decline, generic competition, payer pressure, and regulatory scrutiny.
Marketing Restrictions Increase as Patent Expiration Approaches

During the patent cliff transition period, marketing teams face increasing restrictions from regulators and compliance teams. Companies must ensure that all promotional claims remain strictly within approved labeling and supported by clinical evidence.
Common marketing restrictions during this period include:
- No new promotional claims without new clinical data
- No promotion of unapproved indications
- Increased scrutiny of comparative claims
- Strict fair balance requirements
- Increased review from Medical Legal Regulatory teams
- Greater regulatory attention on digital promotion
- Restrictions on lifecycle claims without supporting evidence
As generic entry approaches, competitors may file legal challenges against promotional claims. Regulators may review promotional materials more closely. Compliance teams often become more conservative because legal risk increases as revenue pressure increases.
This creates a paradox. Marketing teams face increasing revenue pressure at the same time that promotional freedom becomes more restricted.
Lifecycle Marketing Becomes the Primary Strategy
When companies cannot rely on traditional promotion to maintain growth, they turn to lifecycle management strategies. Lifecycle marketing focuses on extending the commercial life of a drug through new indications, new formulations, new delivery systems, and combination therapies.
Common lifecycle strategies include:
- New indication approvals
- Pediatric indications
- Extended-release formulations
- Combination products
- New delivery devices
- Over-the-counter switch if possible
- New dosing schedules
- Real-world evidence publications
These strategies allow companies to differentiate the brand from generic versions and maintain some level of market share after patent expiration.
Lifecycle management often begins years before patent expiration. Marketing teams work closely with research, medical affairs, and regulatory teams to plan these strategies.
Pricing and Market Access Strategy Change During Patent Cliff

As patent expiration approaches, insurers and pharmacy benefit managers prepare to move patients to lower-cost generic alternatives. This creates pricing pressure on the brand drug.
Companies often respond by:
- Offering rebates to insurers
- Negotiating preferred formulary placement
- Launching authorized generics
- Introducing copay assistance programs
- Creating patient loyalty programs
- Offering value-based contracts
During this period, market access strategy becomes more important than advertising. If the brand drug can maintain favorable formulary placement, it can retain some market share even after generic entry.
This means marketing teams must work closely with pricing and market access teams. Commercial strategy becomes more integrated.
The Role of Authorized Generics
One common strategy during patent cliff transitions is launching an authorized generic. An authorized generic is the same drug produced by the brand company but sold under a generic label at a lower price.
This strategy allows the company to:
- Capture part of the generic market
- Compete with other generic manufacturers
- Maintain manufacturing volume
- Protect some revenue after patent expiration
Authorized generics are often launched immediately after patent expiration or sometimes earlier through partnerships with generic manufacturers.
This is not just a pricing strategy. It is a marketing and market positioning strategy.
Physician and Patient Behavior During Patent Cliff
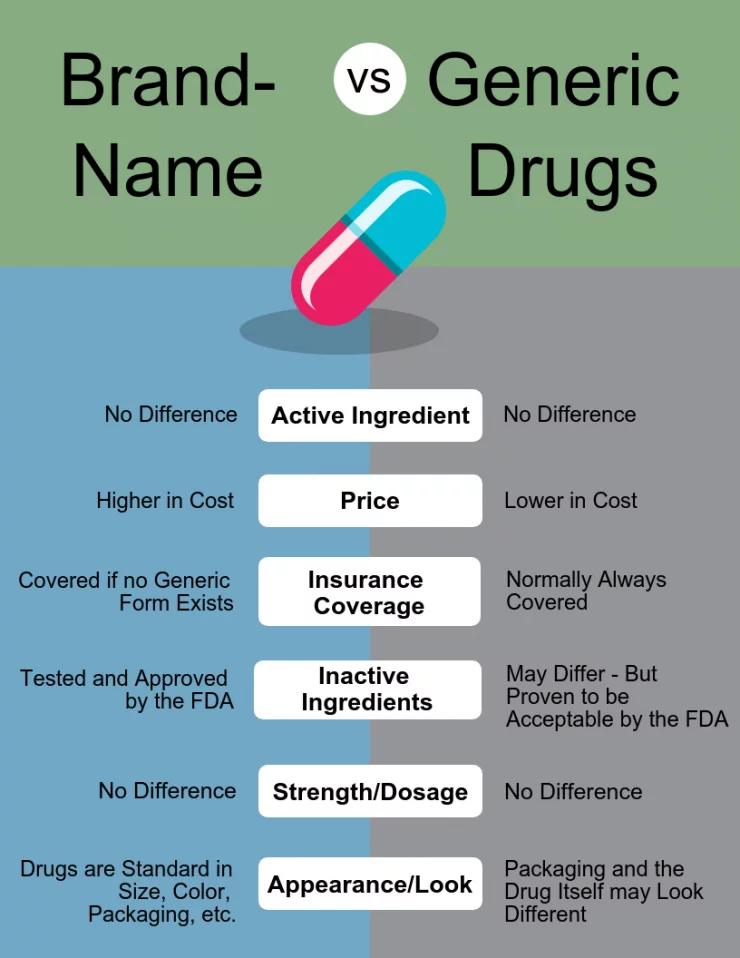
When generics enter the market, several behavioral changes occur:
- Pharmacists substitute generics automatically in many cases
- Insurers encourage generic substitution
- Patients request lower-cost options
- Doctors become less brand loyal
- Hospitals switch to generics for cost reasons
This means brand marketing must shift from awareness to differentiation. Companies must emphasize:
- Reliability and manufacturing quality
- Patient support programs
- Device differences
- Delivery systems
- Clinical data in specific populations
- Real-world evidence
- Brand familiarity and trust
Marketing messages must change because the competitive landscape changes.
Digital Marketing and Brand Loyalty During Patent Cliff
Digital marketing becomes more important during the patent cliff period because companies try to maintain brand loyalty among existing patients and physicians.
Strategies include:
- Patient education programs
- Adherence programs
- Reminder programs
- Brand websites
- Email programs
- Physician education portals
- Real-world evidence publications
- Disease management programs
The goal is not just new prescriptions. The goal is to prevent switching.
Retention becomes more important than acquisition.
Case Studies: What Companies Did During Patent Cliff
Several well-known pharmaceutical companies implemented strategic marketing and lifecycle management during patent cliff transitions.
Examples include:
- Companies developing extended-release versions before patent expiration
- Companies launching combination therapies using the same molecule
- Companies launching biologic improvements before biosimilar entry
- Companies building strong patient support programs to maintain brand loyalty
- Companies negotiating long-term payer contracts before generic entry
These strategies did not stop revenue decline, but they slowed the decline and protected billions in revenue.
Compliance Risk Increases During Revenue Pressure
As revenue declines approach, companies may face internal pressure to maintain sales. This is when compliance risk becomes highest. Promotional claims must remain supported by evidence and within approved labeling.
Many regulatory enforcement actions occur during periods of high commercial pressure because companies may push promotional boundaries.
This is why Medical Legal Regulatory review becomes stricter during patent cliff transitions. Companies must balance commercial pressure with compliance risk.
The Strategic Shift From Growth to Defense

During early product lifecycle, marketing focuses on growth:
- Increasing awareness
- Increasing diagnosis
- Increasing prescriptions
- Expanding indications
During patent cliff transition, marketing focuses on defense:
- Retaining patients
- Retaining formulary placement
- Differentiating the brand
- Launching lifecycle extensions
- Negotiating payer contracts
- Launching authorized generics
- Building brand loyalty
This requires a completely different marketing strategy. Many marketing teams are trained for growth marketing but not trained for defensive marketing.
The Most Important Strategic Question During Patent Cliff
When patent expiration approaches, companies must ask a critical question.
What percentage of patients will remain on the brand after generic entry, and what strategy will keep them on the brand?
This question determines lifecycle strategy, pricing strategy, patient support programs, physician engagement strategy, and digital marketing strategy.
Patent cliff management is not just a legal or regulatory issue. It is a commercial strategy issue that requires coordination across marketing, medical affairs, pricing, market access, regulatory, and manufacturing teams.
The companies that manage patent cliff transitions successfully do not try to stop generic competition. They try to control how fast revenue declines and how much market share they can retain.
Patent cliffs are inevitable. Revenue collapse is not entirely inevitable. Strategy determines how steep the fall will be.
References
FDA Orange Book – Approved Drug Products with Therapeutic Equivalence Evaluations
https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-data-files
Evaluate Pharma Patent Cliff Report
https://www.evaluate.com/thought-leadership/pharma/patent-cliff-report
IQVIA Institute Report on Loss of Exclusivity and Lifecycle Management
https://www.iqvia.com/insights/the-iqvia-institute
Deloitte Report – Managing Pharmaceutical Patent Expiry
https://www2.deloitte.com
McKinsey & Company – Pharma Lifecycle Strategy Report
https://www.mckinsey.com/industries/life-sciences
Harvard Business Review – Strategies for Managing Patent Expiry
https://hbr.org

