
Drug price transparency laws are not just policy changes. They are forcing pharmaceutical companies to change how they communicate value, pricing, and clinical benefit. For decades, pharmaceutical pricing operated in a system where list prices, rebates, insurance negotiations, and out-of-pocket costs remained largely invisible to patients. That environment is changing. Governments, insurers, employers, and patients are demanding price visibility, and this shift is transforming pharmaceutical marketing communication strategy.
If you work in pharmaceutical marketing, you can no longer communicate only clinical benefits. You must communicate economic value, affordability, access, and cost justification. Price transparency is turning pricing communication into a core part of marketing strategy.
The companies that adapt their communication strategy to this new environment will maintain market access and brand trust. The companies that continue using old promotional models will face payer resistance, public criticism, and slower adoption.
The Policy Shift That Changed Pricing Communication

Over the past decade, policymakers in the United States and other countries have introduced multiple price transparency laws aimed at controlling healthcare costs and improving patient awareness. These laws require hospitals, insurers, and in some cases pharmaceutical companies to disclose pricing information.
Key policy developments include:
- The U.S. Sunshine Act, which increased transparency around payments to physicians
- CMS hospital price transparency rules requiring hospitals to publish prices
- Insurer transparency rules requiring disclosure of negotiated rates
- State-level drug price transparency laws requiring justification for price increases
- Federal proposals requiring disclosure of drug prices in advertisements
These policies aim to expose pricing structures that were previously difficult for patients and employers to understand. Once pricing becomes visible, marketing communication must change. You cannot promote a drug without addressing cost questions when the price is publicly available.
Why Price Transparency Changes Marketing Strategy

When pricing becomes visible, value communication becomes essential. Pharmaceutical companies must explain why a drug costs what it costs and what value it delivers compared to existing treatments.
Marketing communication must now address:
- Clinical benefit
- Cost effectiveness
- Impact on hospitalizations
- Impact on disease progression
- Quality of life improvements
- Productivity benefits
- Caregiver burden reduction
- Long-term healthcare cost savings
This type of communication is often called value communication, and it sits at the intersection of marketing, medical affairs, and market access.
If your marketing team cannot communicate economic value, your payer negotiations become harder and your brand perception becomes weaker.
The Difference Between Price and Cost in Pharmaceutical Communication
One of the biggest communication challenges in pharmaceutical marketing is explaining the difference between list price and actual cost to patients. Many patients see high list prices reported in the media and assume that is what they will pay out of pocket. In reality, insurance coverage, rebates, copay assistance, and patient assistance programs often reduce patient cost significantly.
Marketing communication strategies must clearly explain:
- List price
- Net price after rebates
- Insurance coverage scenarios
- Copay assistance programs
- Patient assistance programs
- Free drug programs for eligible patients
If companies do not explain these clearly, public perception focuses only on high list prices.
Communication Strategy for Different Stakeholders
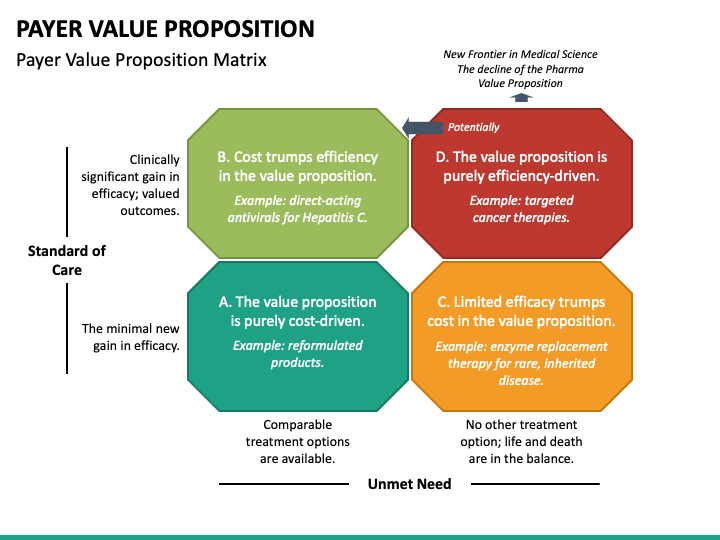
Drug price transparency affects different stakeholders in different ways, and communication strategy must be tailored for each group.
Communication with Patients
Patients want to know:
- How much will I pay
- Is there financial assistance
- Is the drug covered by insurance
- Is there a cheaper alternative
- What happens if I cannot afford treatment
Communication with Physicians
Physicians want to know:
- Insurance coverage
- Prior authorization requirements
- Copay levels
- Patient assistance programs
- Clinical benefit compared to cost
Communication with Payers
Payers want to know:
- Cost effectiveness
- Budget impact
- Comparative effectiveness
- Real-world outcomes
- Long-term cost savings
Communication with Policymakers
Policymakers want to know:
- Why the drug is priced at a certain level
- How the drug reduces healthcare system costs
- How innovation costs are recovered
- How pricing compares globally
This means pharmaceutical marketing communication is no longer one message. It is multiple value narratives tailored to each stakeholder.
Health Economics and Outcomes Research Is Now a Marketing Tool
Health economics and outcomes research, often called HEOR, has become one of the most important tools in pharmaceutical marketing communication. HEOR studies demonstrate the economic value of a drug, not just clinical efficacy.
HEOR evidence may include:
- Cost-effectiveness analysis
- Budget impact analysis
- Quality-adjusted life years
- Hospitalization reduction data
- Productivity improvement data
- Real-world evidence
- Long-term outcome data
Marketing teams now use HEOR data in:
- Payer presentations
- Value dossiers
- Physician education materials
- Policy discussions
- Market access negotiations
In a price transparency environment, clinical data alone is not enough. Economic value data becomes essential.
Real-World Evidence Is Changing Pricing Discussions
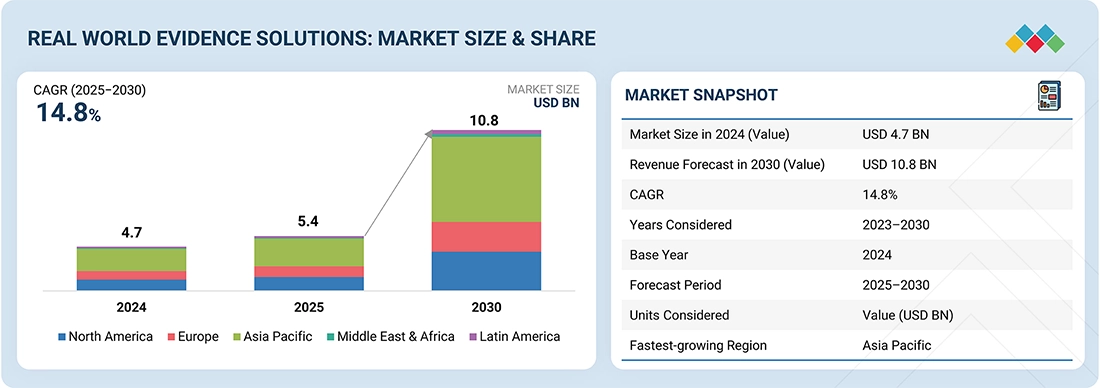
Real-world evidence includes data collected after a drug is launched, such as patient outcomes, hospitalizations, adherence rates, and long-term effectiveness. This data helps companies demonstrate whether a drug delivers value in real clinical practice.
Real-world evidence is increasingly used in:
- Value-based pricing agreements
- Outcomes-based contracts
- Payer negotiations
- Policy discussions
- Marketing communication
If a drug reduces hospitalizations or prevents disease progression, companies can use that data to justify pricing and maintain coverage.
Value-Based Pricing and Outcomes-Based Contracts
Some pharmaceutical companies now enter value-based agreements where payment depends on patient outcomes. If the drug does not perform as expected, the company provides rebates or refunds.
This model changes marketing communication because companies must communicate expected outcomes clearly and support those claims with data.
Value-based pricing communication focuses on:
- Expected clinical outcomes
- Cost savings from avoided complications
- Long-term health system savings
- Risk-sharing agreements with payers
This is very different from traditional promotional messaging.
The Risk of Poor Price Communication
If pharmaceutical companies do not communicate pricing and value effectively, several risks appear:
- Negative media coverage
- Political pressure
- Payer restrictions
- Patient distrust
- Lower adherence
- Brand reputation damage
Price transparency means pricing is now part of brand perception.
The Future of Pharmaceutical Marketing Communication
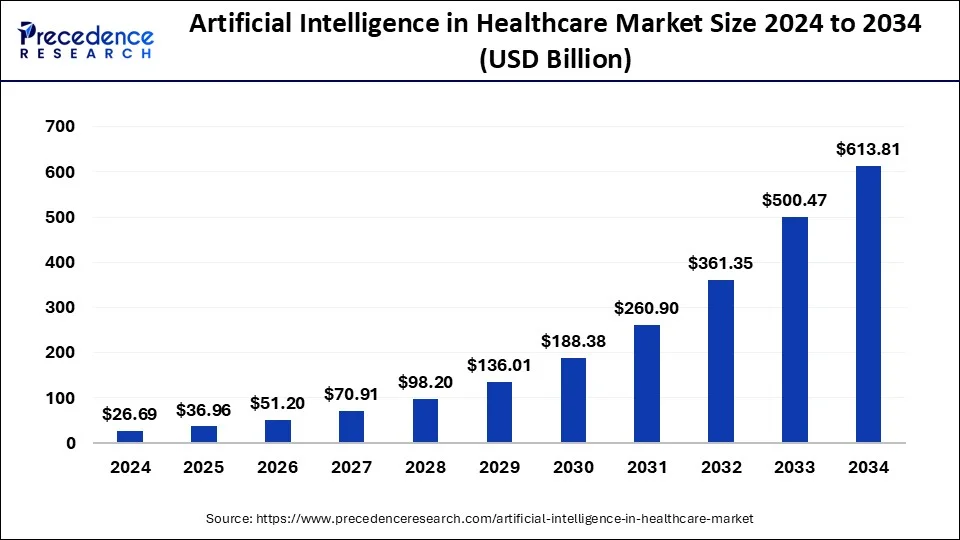
Drug price transparency laws are pushing pharmaceutical marketing toward value communication, data communication, and economic storytelling supported by evidence.
Future pharmaceutical marketing communication will focus on:
- Clinical value
- Economic value
- Real-world outcomes
- Patient affordability
- Access and coverage
- Value-based pricing
- Transparency reports
- Digital patient education
- Personalized cost calculators
The companies that communicate value clearly will maintain access and trust. The companies that communicate only clinical benefits without addressing price will face resistance from payers, policymakers, and patients.
The Strategic Question for Pharmaceutical Companies
If your drug price becomes public tomorrow, can you clearly explain why the drug costs what it costs and what value it delivers to patients, insurers, and the healthcare system?
If the answer is no, then your marketing communication strategy is not ready for the transparency era.
Drug price transparency laws are not just policy changes. They are forcing pharmaceutical companies to become better at explaining value, not just promoting products.
That is a communication challenge, a market access challenge, and a marketing strategy challenge at the same time.
References
CMS Hospital Price Transparency Rule
https://www.cms.gov/hospital-price-transparency
U.S. Sunshine Act Overview
https://www.cms.gov/openpayments
IQVIA Report on Drug Pricing and Market Access
https://www.iqvia.com/insights/the-iqvia-institute
Health Affairs – Drug Price Transparency Policy Analysis
https://www.healthaffairs.org
Deloitte – Pharmaceutical Pricing and Market Access Trends
https://www2.deloitte.com
McKinsey – Pharmaceutical Pricing and Value Communication Report
https://www.mckinsey.com

